pBT2
pBT2
编号 | 载体名称 |
北京华越洋VECT75325 | pBT2 |
pBT2载体基本信息:
载体名称: | pBT2 |
质粒类型: | 金黄色葡萄球菌基因敲除载体,金葡菌基因敲除载体 |
高拷贝/低拷贝: | 低拷贝 |
克隆方法: | 限制性内切酶,多克隆位点 |
启动子: | -- |
载体大小: | 6.97kb |
5' 测序引物及序列: | -- |
3' 测序引物及序列: | -- |
载体标签: | -- |
载体抗性: | 氨苄青霉素 |
筛选标记: | 氯霉素 |
克隆菌株: | DH5α 等 |
宿主细胞(系): | 金黄色葡萄球菌等革兰氏阳性菌 |
备注: | 该载体是大肠杆菌-金黄色葡萄球菌穿梭载体,低拷贝,在大肠中抗性为氨苄,在金葡菌中抗性为氯霉素。该质粒具有温度敏感性。 |
稳定性: | 稳表达 |
组成型/诱导型: | 组成型 |
病毒/非病毒: | 非病毒 |
pBT2载体质粒图谱和多克隆位点信息:
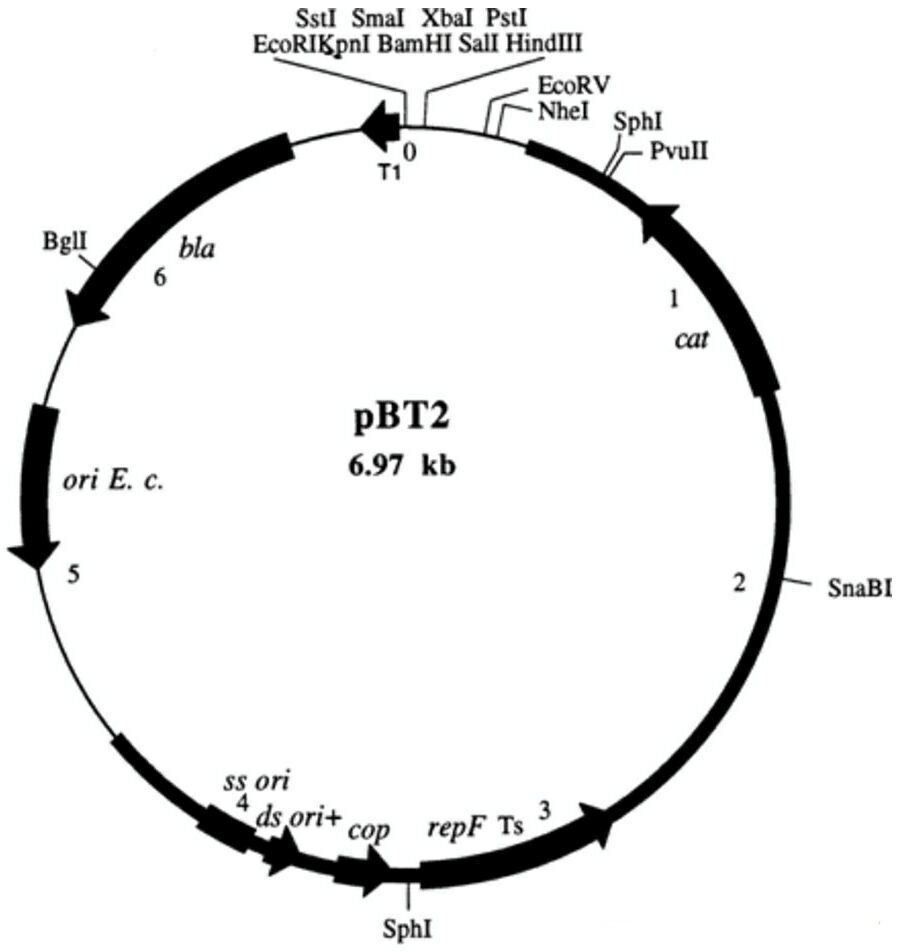
pBT2载体简介:
Protocols for gene deletion in Staphylococcus aureus Nov. 1, 2007
Preparation of competent Staphylococcus aureus cells
1. Remove Staphylococcus aureus cells from the vial with a sterile toothpick or inoculation loop, and streak it out on LB agar.
2. Incubate at 37°C overnight.
3. Pick a single colony and inoculate it in 5-10 ml of LB. Grow at 37°C overnight.
4. Add 1 ml overnight culture to 100 ml LB medium in a 500 ml flask, and shake at 37°C until an OD600 of 0.4 is reached (approximately 90–120 min).
5. Cool the culture on ice for 5 min, and transfer the culture to a sterile, round-bottom centrifuge tube.
6. Collect the cells by centrifugation at low speed (5-10 min, 2500 x g, 4°C).
7. Discard the supernatant carefully. Always keep the cells on ice.
8. Resuspend the cells gently in 0.5 M sucrose (10-15 ml for a 100 ml culture) at 4°C and keep the suspension on ice for additional 5 min.
9. Collect the cells by centrifugation (5 min, 2500 x g, 4°C).
10. Discard the supernatant carefully. Repeat step 8 and 9.
11. Resuspend the cells carefully in 1 ml ice-cold 0.5 M sucrose and keep the suspension on ice for 15 min.
12. Prepare aliquots of 100–200 μl in sterile microcentrifuge tubes and freeze in liquid nitrogen. Store the competent cells at –70°C.
Construction of deletion vector
1. PCR amplify a 400 bp fragment upstream and a 400 bp fragment downstream of the target gene.
2. PCR amplify the ermB (Em resistance marker) from pECI.
3. Digest the three fragments, ligate, and PCR amplify the ligated product.
4. Purify the PCR product, double digest it, and ligate it into pBT2.
5. Transform the ligated product into E. coli.
6. Pick clones that can grow on the LB plate containing Em (100 mg/ml), purify the plasmid and digest it.
7. If the result of enzyme digestion is correct, get further confirmation by sequencing.
Procedure for electroporation
1. Mix 500 ng of plasmid DNA with electrocompetent Staphylococcus aureus cells and place them in a Gene Pulser cuvette with a 0.2 cm electrode gap.
2. The settings for electroporation are as follows: Voltage, 2.5 kV; capacitor, 50 μF; resistance, 200 ohms.
3. After electroporation the cells are immediately placed in 400 μl of TSB with shaking (200-220 rpm, 37°C) for 1h. Plate the cells on Em-containing medium and incubate at 37°C.
Modify deletion vector
1. Before transform the plasmid into Staphylococcus aureus NCTC8325, the plasmid should be transformed into Staphylococcus aureus RN4220.
2. Pick clones, after overnight growth, extract the plasmid.
3. Then the plasmid is modified and can’t be digested by restriction enzyme system of NCTC8325.
Deletion of target gene
1. Extract plasmid from RN4220, transform it into NCTC8325.
2. Pick up clones, incubate in B-medium, 30°C, grow to late-stationary phase, then change temperature to 40°C, and grow overnight.
3. 1: 100 dilute the culture into fresh B-medium, and grow overnight.
4. Follow step 3, spread 1 μl overnight culture (diluted into 100 μl) on agar plate (containing Em 2.5 mg/ml). Screen clones which are Em-resistant and Cm-sensitive.
5. Repeat step 4 until Em-resistant, Cm-sensitive clones are found.
6. Extract genome DNA of these clones, use PCR for further check.
pBT2载体其他相关的其他类型载体:


